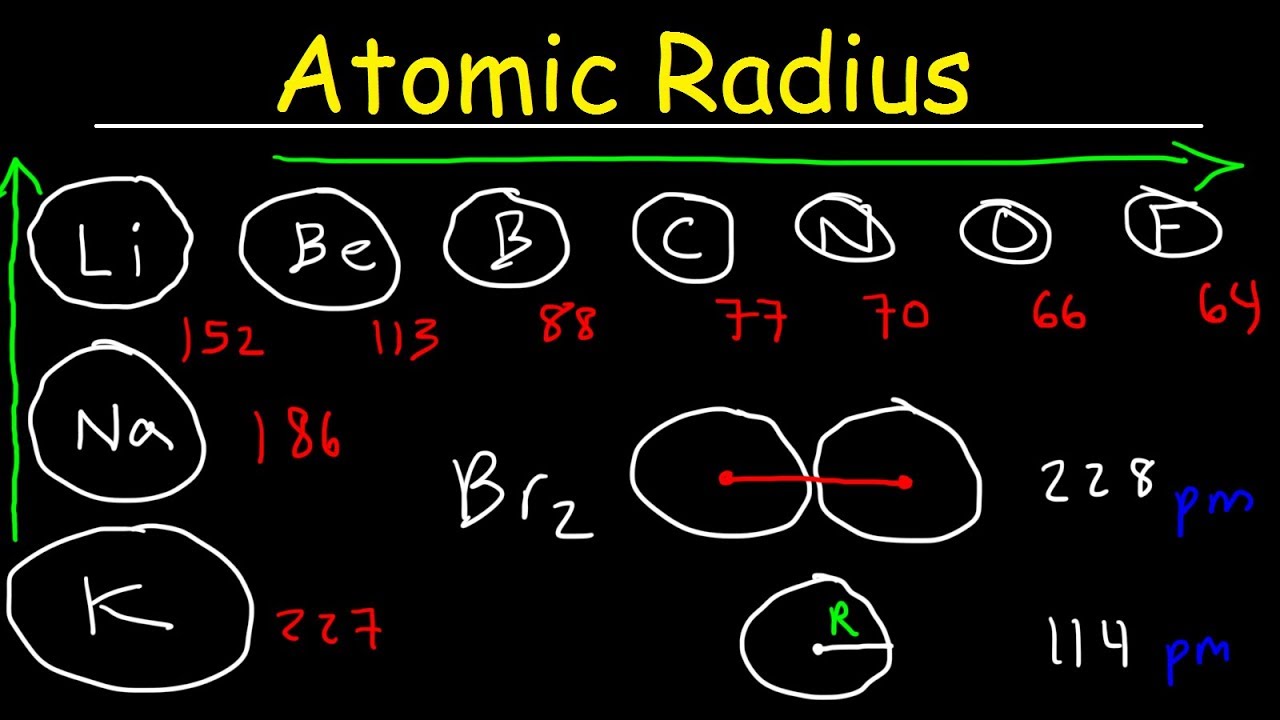
This chemistry video tutorial provides a basic introduction into atomic radius which is one of the four main periodic table trends you need to know. Atomic radius increases as you down a group and to the left across the periodic table. Atomic radius decreases with effective nuclear charge but increases with each successive energy level added to an atom as you go down a group.
Ionization Energy:
[ Ссылка ]
Electron Affinity:
[ Ссылка ]
Atomic Radius:
[ Ссылка ]
Bond Energy & Bond Length:
[ Ссылка ]
Electronegativity:
[ Ссылка ]
Periodic Trends:
[ Ссылка ]
__________________________________
Polar & Nonpolar Covalent Bonding:
[ Ссылка ]
Bond Polarity & Dipole Moment:
[ Ссылка ]
Ionic Radius:
[ Ссылка ]
Lattice Energy:
[ Ссылка ]
Born Haber Cycle:
[ Ссылка ]
Bond Energy Calculations:
[ Ссылка ]
___________________________________
Lewis Structures - Mega Review:
[ Ссылка ]
Final Exams and Video Playlists:
[ Ссылка ]
Full-Length Videos and Worksheets:
[ Ссылка ]