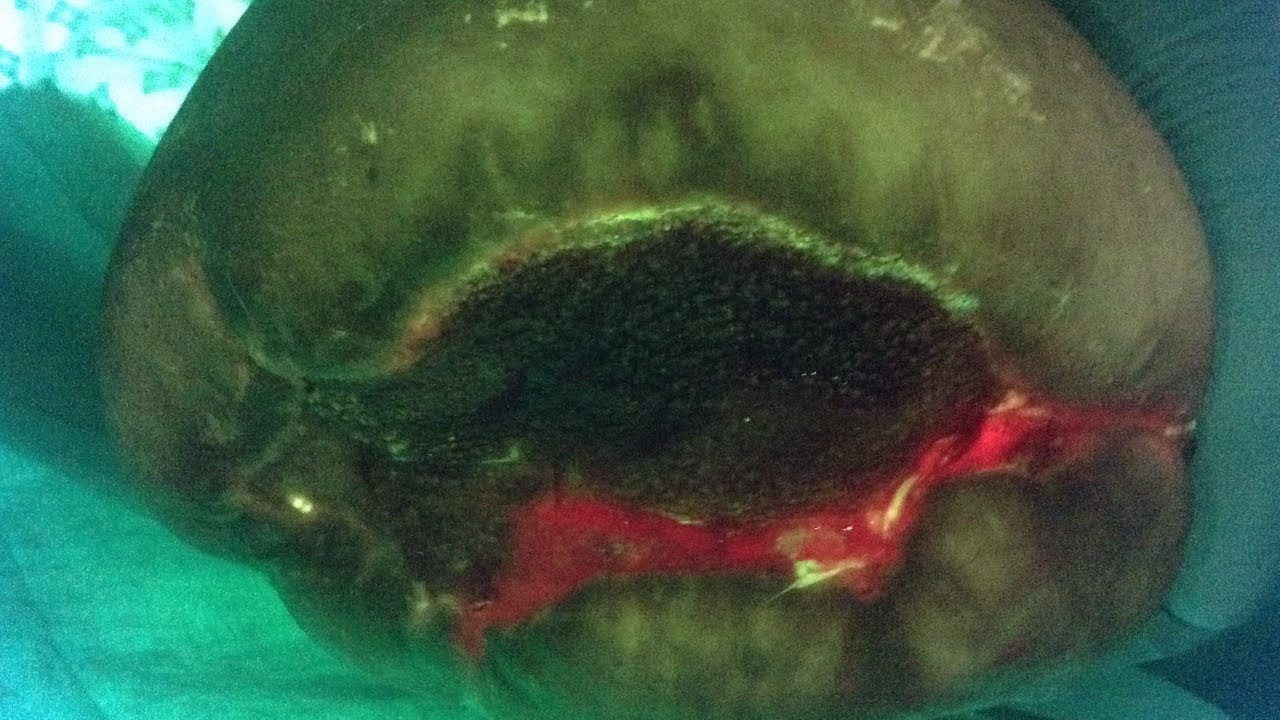
In this video, the clinician used the MolecuLight i:X Imaging Device in Fluorescence Imaging Mode to visualize bacterial contamination of a limb amputation in real-time. Red fluorescence indicates presence of bacteria at the surgical site. Swabs later confirmed E. coli and P. mirabilis. As a result, further wash-out was performed prior to closure.
Of all surgical wounds, the highest rate of surgical site infection occurs with lower limb amputations1. Infection complications include rupture of the suture line, cellulitis, delayed healing and further surgery. It is vital to aggressively monitor amputation wounds for clinical signs and symptoms of infection and the MolecuLight i:X offers invaluable insights for clinicians.
Reference:
1. Surgical Site Infection Surveillance Service. CDR Weekly 2006; 14(21): 1-5.
Video provided by Lt Col Steven Jeffery, The Royal Centre for Defence Medicine, Birmingham, UK
MolecuLight Clinical Case 0014.
Inquiries:
info@moleculight.com
Website:
[ Ссылка ]
Twitter: @moleculight
The MolecuLight i:X™ Imaging Device is approved by Health Canada (Medical License #95784) and has CE marking (Certificate #G1160292355002) for sale in Canada and the European Union. The MolecuLight i:X™ Imaging Device is not available in the US.
MolecuLight® and Look to Heal® are Registered Trademarks in Canada, the US, and the EU.