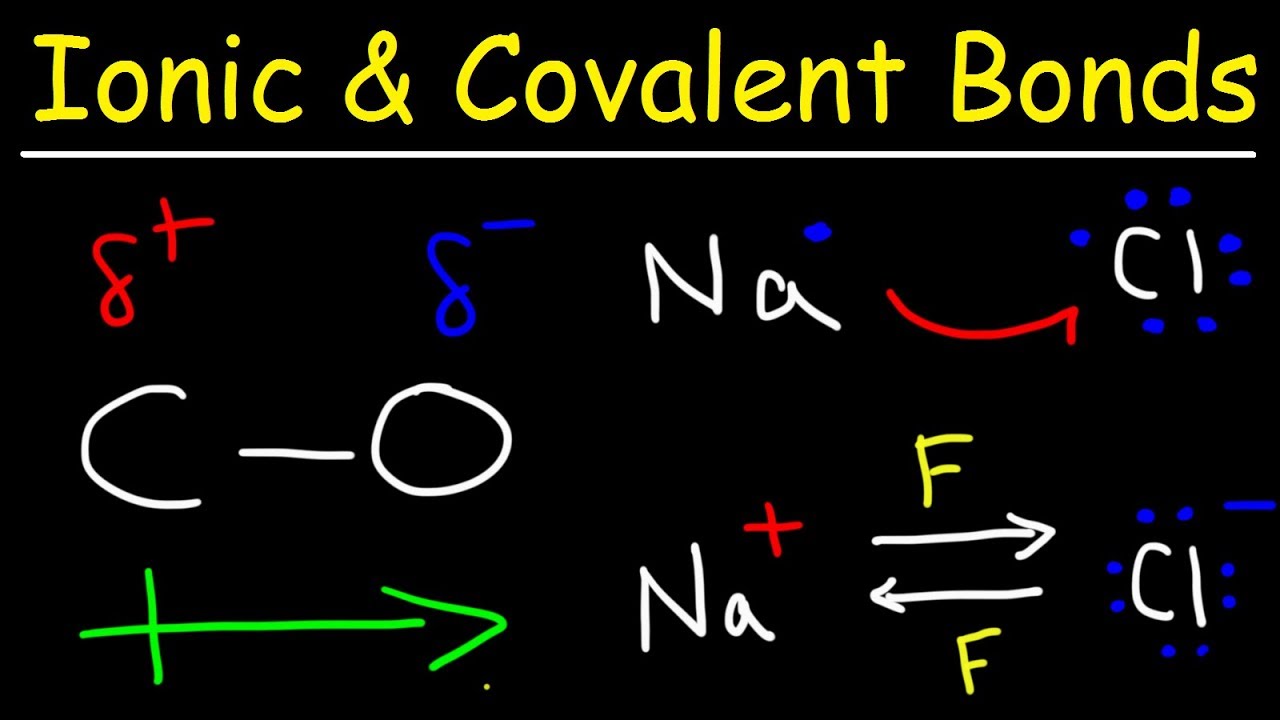
This organic chemistry video tutorial explains how to identify a bond as an ionic bond, polar covalent bond, or a nonpolar covalent bond. Ionic bonds usually consist of metals and nonmetals where as covalent bonds consists of nonmetals. In a nonpolar covalent bond, electrons are shared equally and the electronegativity difference between the two atoms is 0.4 or less. For polar covalent bonds, the electrons are shared unequally between the two atoms and the electronegativity difference is defined to be 0.5 or more.
Organic Chemistry - Basic Introduction:
[ Ссылка ]
Which Bond Is More Polar?
[ Ссылка ]
How To Draw Lewis Structures:
[ Ссылка ]
Condensed Structures to Skeletal Structures:
[ Ссылка ]
Functional Groups Review:
[ Ссылка ]
Primary, Secondary, & Tertiary Functional Groups:
[ Ссылка ]
_________________________________
How To Calculate Formal Charge:
[ Ссылка ]
Finding Lone Pairs Using Formal Charge:
[ Ссылка ]
Dipole Moment & Electronegativity:
[ Ссылка ]
Predicting Bond Angles:
[ Ссылка ]
Valence Bond Theory:
[ Ссылка ]
Hybridization of Atomic Orbitals:
[ Ссылка ]
_______________________________
Bond Strength and Bond Length:
[ Ссылка ]
Orbital Overlap and Bond Length:
[ Ссылка ]
Organic Chemistry PDF Worksheets:
[ Ссылка ]
Organic Chemistry Exam 1 Playlist:
[ Ссылка ]
Full-Length Videos and Worksheets:
[ Ссылка ]