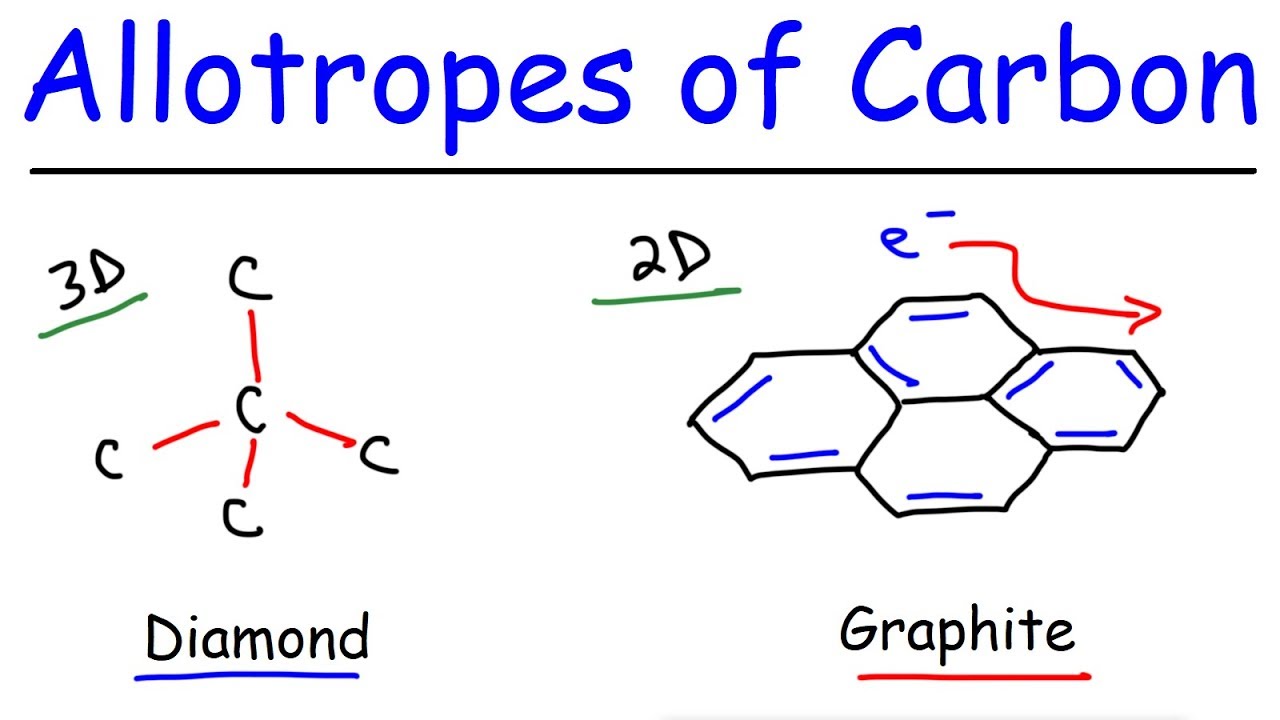
This chemistry video tutorial provides a basic introduction into some of the different types of allotropes of carbon. These include graphite, diamond, graphene, and fullerenes such as carbon nanotubes and buckminister fullerenes also known as bucky balls. Graphite forms a 2D structure consisting of sp2 hybridized carbon atoms. Each layer of graphite is held by weak van der waal forces. Graphite conducts electricity due to the delocalization of its pi electrons. Both graphite and diamond form large network covalent structures. However, diamond does not conduct electricity due to its localized electrons. It is however, an excellent conduct of heat.
How To Draw Lewis Structures: [ Ссылка ]
VSEPR Theory:
[ Ссылка ]
Molecular Geometry:
[ Ссылка ]
Lewis Dot Structures:
[ Ссылка ]
Lewis Structures of Ionic Compounds:
[ Ссылка ]
_________________________________
Octet Rule Exceptions:
[ Ссылка ]
Resonance Structures:
[ Ссылка ]
Polar and Nonpolar Molecules:
[ Ссылка ]
Formal Charge Calculations:
[ Ссылка ]
Lewis Structures - Mega Review:
[ Ссылка ]
________________________________
Hybridization of Atomic Orbitals:
[ Ссылка ]
Molecular Orbital Theory:
[ Ссылка ]
Dipole Dipole Forces of Attraction:
[ Ссылка ]
Hydrogen Bonding:
[ Ссылка ]
Unit Cell Chemistry:
[ Ссылка ]
_________________________________
Final Exams and Video Playlists:
[ Ссылка ]
Full-Length Videos and Worksheets:
[ Ссылка ]