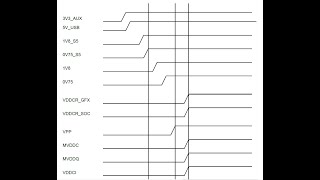
This video introduces the Europan medical device regulations, in particular the Medical Device Regulation MDR, the conformity assessment procedures (sometimes referred to as "certification process") and the medical device classification.
It explains when an ISO 13485 certified quality management system is required and which are the most relevant documents manufacturers have to compile
You find the checklist mentioned in the video and additional information in our starter-kit: [ Ссылка ]








![[4K USA] HOME vacuuming | TRY ON HAUL Transparent White DRESS with Janet](https://s2.save4k.ru/pic/T9VlHpT5WPQ/mqdefault.jpg)




























































![РЕКЛАМА | МТС - МОЙ БЕЗЛИМИТИЩЕ [1]](https://s2.save4k.ru/pic/avfN_j85PA4/mqdefault.jpg)


![Lenovo G70-80 - disassemble [4K]](https://s2.save4k.ru/pic/370FvGa6f5s/mqdefault.jpg)