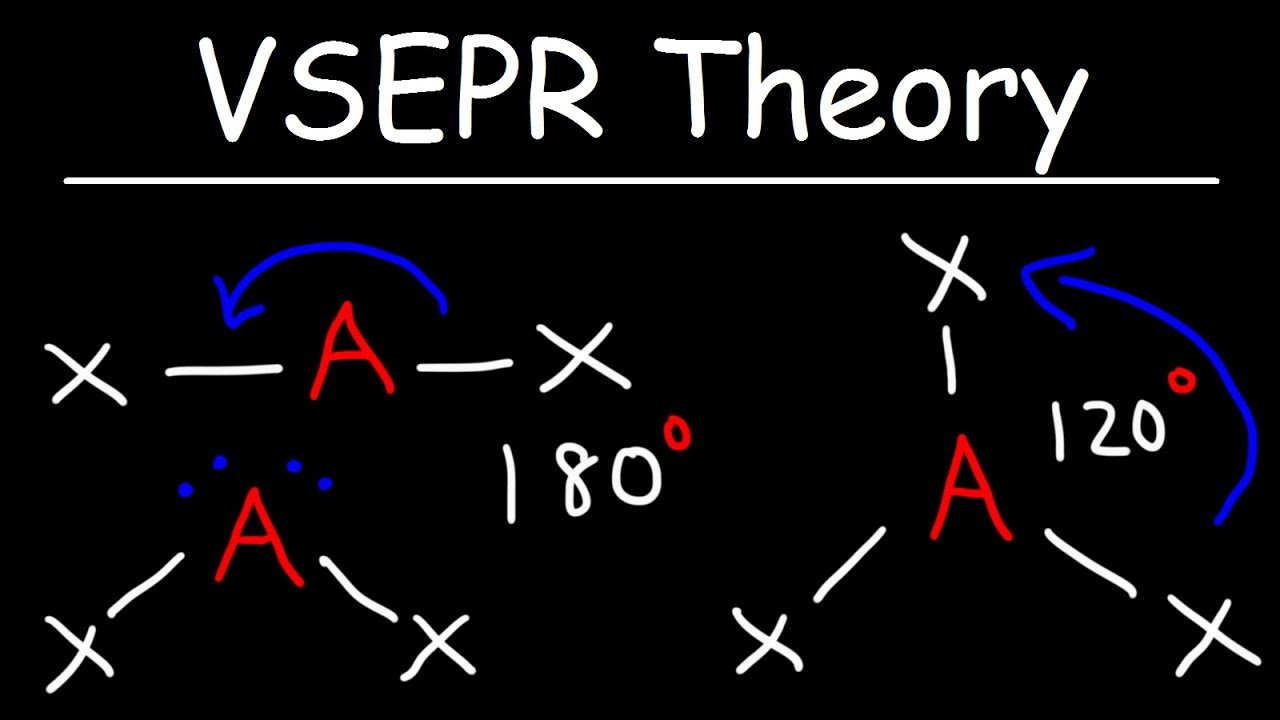
This chemistry video tutorial provides a basic introduction into VSEPR theory and molecular structure. It contains examples and practice problems of drawing lewis structures along with the correct molecular geometry. Structures include the tetrahedral shape, bent, linear, trigonal planar, and trigonal pyramidal molecular geometry as well as their bond angles.
How To Draw Lewis Structures:
[ Ссылка ]
VSEPR Theory:
[ Ссылка ]
Molecular Geometry:
[ Ссылка ]
Lewis Dot Structures:
[ Ссылка ]
Lewis Structures of Ionic Compounds:
[ Ссылка ]
_________________________________
Octet Rule Exceptions:
[ Ссылка ]
Resonance Structures:
[ Ссылка ]
Polar and Nonpolar Molecules:
[ Ссылка ]
Formal Charge Calculations:
[ Ссылка ]
Lewis Structures - Mega Review:
[ Ссылка ]
________________________________
Hybridization of Atomic Orbitals:
[ Ссылка ]
Molecular Orbital Theory:
[ Ссылка ]
Dipole Dipole Forces of Attraction:
[ Ссылка ]
Hydrogen Bonding:
[ Ссылка ]
Unit Cell Chemistry:
[ Ссылка ]
_________________________________
Final Exams and Video Playlists:
[ Ссылка ]
Full-Length Videos and Worksheets:
[ Ссылка ]