The Periodic Table provides a list of all of the known Elements. In this video we’ll look at the difference between the term elements and atoms.
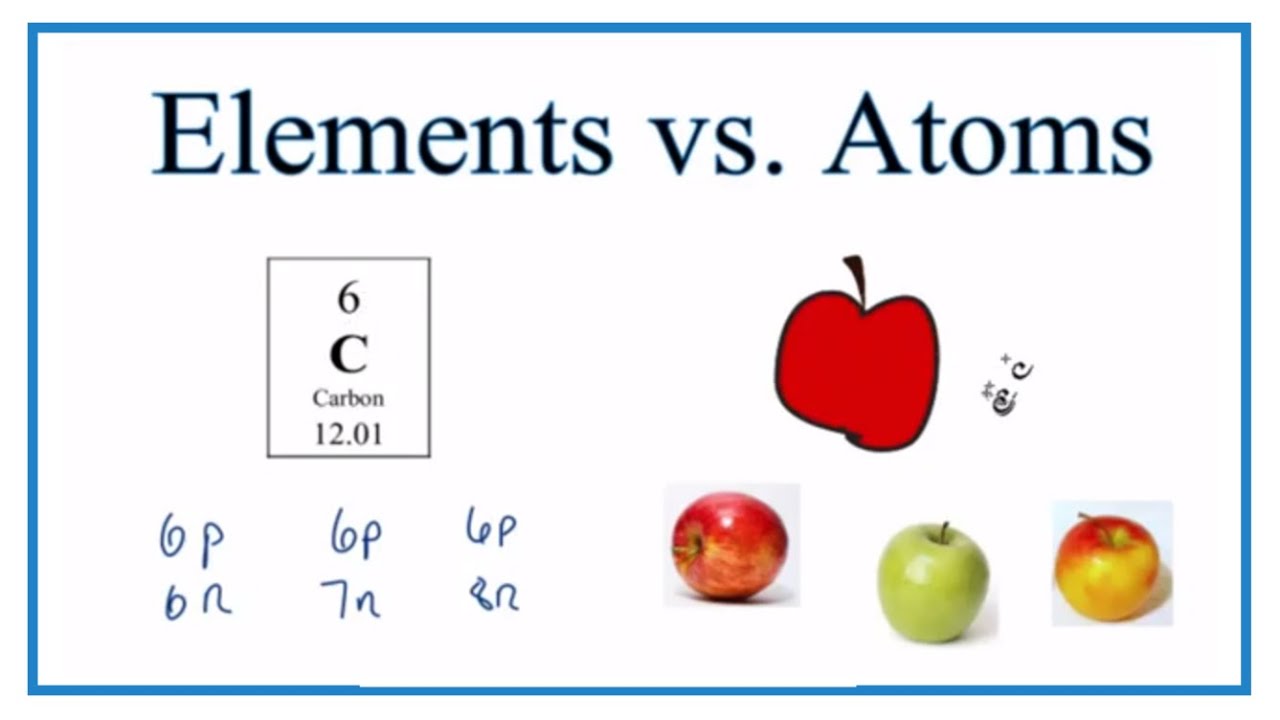
An atom is an individual particle made up of protons, neutrons, and electrons. We could say “the element Carbon is made up of atoms with six protons and either six, seven, or eight neutrons. ”
Atoms that have the same atomic number are considered to be the same element. For example, Carbon (C), Gold (Au), or Hydrogen (H). We say “element” we are referring to a category of atoms with the same number of protons. The number of neutrons may differ. We call these isotopes of that element.
Help Videos:
Intro to Isotopes: [ Ссылка ]
Protons, Neutrons, & Electrons: [ Ссылка ]
Finding Charge for Ions: [ Ссылка ]
PhET Web App: [ Ссылка ]
Images:
Gala Apple: [ Ссылка ]
Granny Smith Apple: [ Ссылка ]
Jazz Apple: [ Ссылка ]











![JavaScript - Полный Курс JavaScript Для Начинающих [11 ЧАСОВ]](http://i.ytimg.com/vi/CxgOKJh4zWE/mqdefault.jpg)






























































