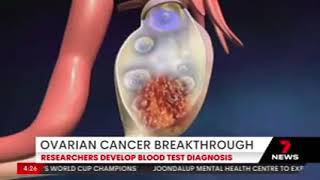
A new blood test is now being developed to improve ovarian cancer diagnosis, with the potential to reduce unnecessary surgery leading to better health outcomes, saving time, stress and money for patients and the healthcare system.
Hudson Institute of Medical Research, funded by the Ovarian Cancer Research Foundation (OCRF), has developed a new diagnostic test for ovarian cancer, which is now being commercialised by Australian company Cleo Diagnostics Ltd.
The blood tests will be underpinned by a patented novel biomarker, CXCL10, which is produced early and at high levels by ovarian cancers, but not in non-malignant disease. CXCL10 was first identified at Hudson Institute by Research Group Head of the Ovarian Cancer Biomarkers Research Group, Dr Andrew Stephens, who is also now Cleo’s Chief Scientific Officer.
For more information contact us:
Hudson Institute communications
t: + 61 3 8572 2761
e: communications@hudson.org.au
w: www.hudson.org.au