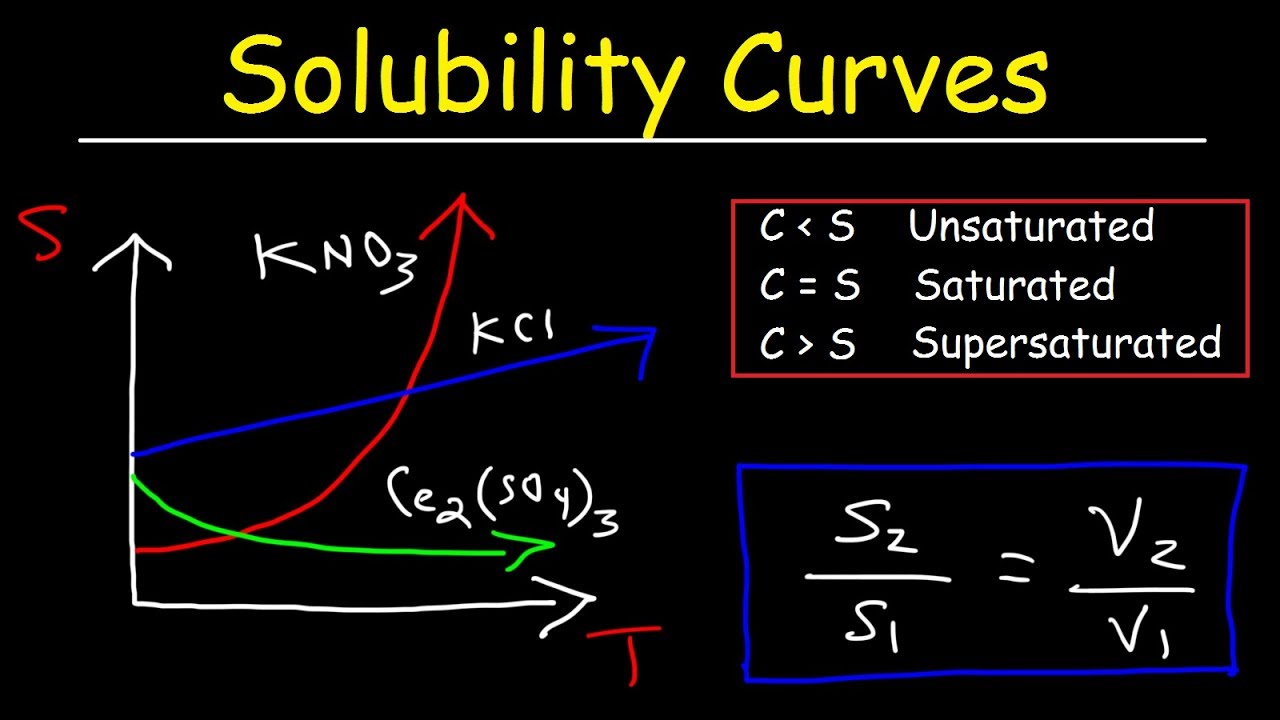
This chemistry video tutorial provides a basic introduction into solubility curves. It contains plenty of examples and practice problems on calculating the solubility of an ionic compound at a given temperature and volume. It explains how to determine the temperature at which two substances have the same solubility in addition to determining if a solution is saturated, unsaturated, or supersaturated under certain conditions.
Heating Curve of Water:
[ Ссылка ]
Heating Curve Chemistry Problems:
[ Ссылка ]
Final Temperature - Ice Water Mixture:
[ Ссылка ]
Molarity, Molality, Density, & Mass %:
[ Ссылка ]
Normality & Equivalent Weight:
[ Ссылка ]
_________________________________
PPM and PPB Concentrations:
[ Ссылка ]
How To Convert PPM to Molarity:
[ Ссылка ]
Enthalpy of Solution & Hydration:
[ Ссылка ]
Solubility Vs Concentration:
[ Ссылка ]
Solubility Curves:
[ Ссылка ]
___________________________________
Henry's Law & Gas Solubility:
[ Ссылка ]
Vapor Pressure & Clausius Equation:
[ Ссылка ]
Raoult's Law - Vapor Pressure:
[ Ссылка ]
Colligative Properties:
[ Ссылка ]
Chemical Kinetics Initial Rate Method:
[ Ссылка ]
_________________________________
Final Exams and Video Playlists:
[ Ссылка ]
Full-Length Videos and Worksheets:
[ Ссылка ]
Chemistry PDF Worksheets:
[ Ссылка ]