directory of Chem Help ASAP videos: [ Ссылка ]
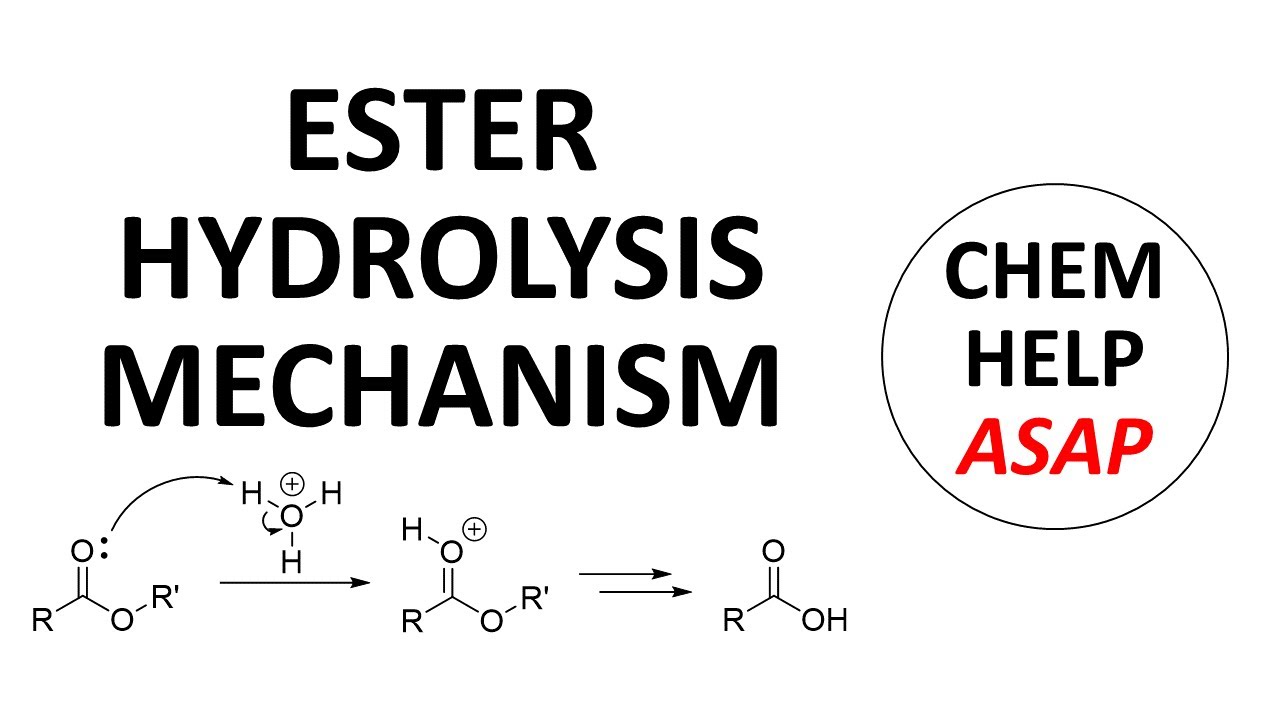
The mechanism for the acid-catalyzed hydrolysis of esters is very similar to the other acid derivatives. The reaction starts with protonation of the ester carbonyl. Water attacks the carbonyl in an addition reaction. After two proton transfers, the OR' group can be lost as an alcohol. A final deprotonation with water gives the carboxylic acid product. Amides, nitriles, and acid chlorides follow a very similar reaction to give carboxylic acids.
acid-catalyzed mechanism of ester hydrolysis
Теги
organic chemistryacid derivativeacid catalyzed hydrolysis of estermechanism of ester hydrolysisacid catalyzed hydrolysishydrolysis of esters mechanismacid derivative hydrolysishydrolysis of acid derivativesChem Help ASAPchemhelpasapleah4scichemistryorganicvideoprofessor dave explainsprofessordaveexplainskhankhan academykhan organicmcatmcat organicester hydrolysis mechanismacid catalyzed ester hydrolysis mechanismmastertutor