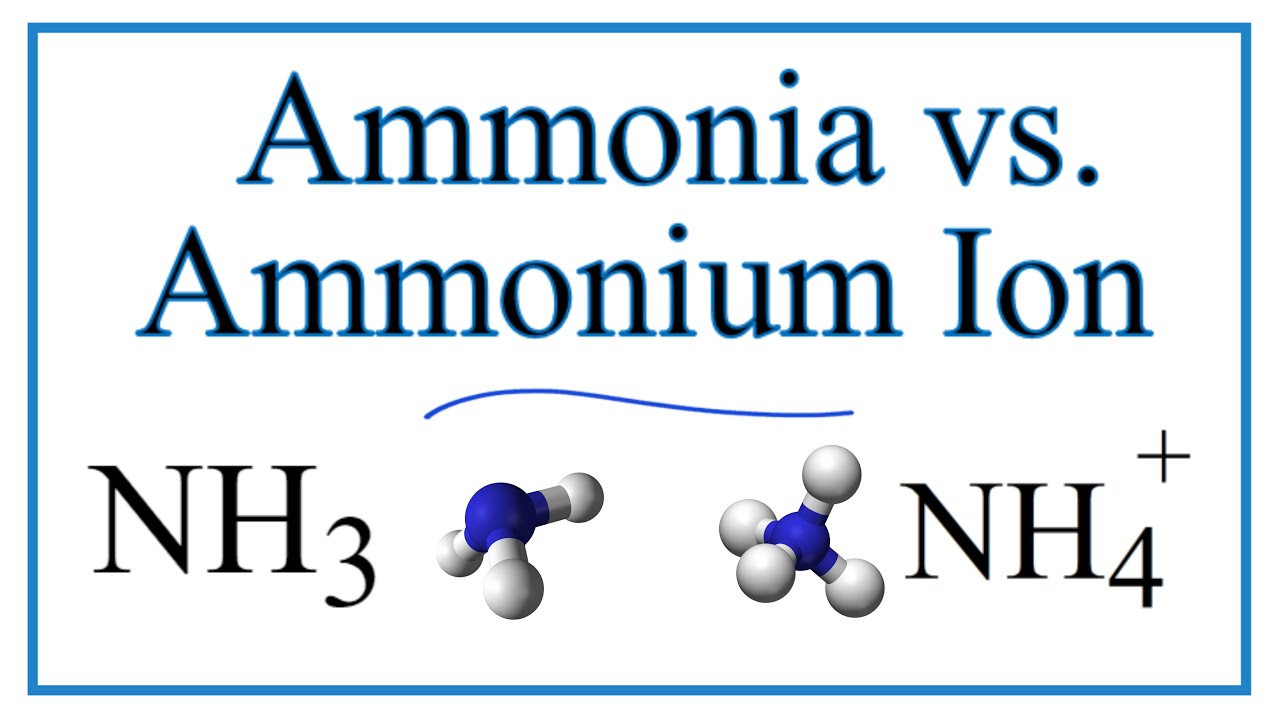
A brief explanation of the differences between Ammonia (NH3) and the Ammonium Ion (NH4+) including Lewis structures, molecular geometry, and bond angles.
In comparing and contrasting Ammonia and the Ammonium ion:
• Ammonia is a strong smelling substance. The Ammonium ion has no odor.
• Both Ammonia and the Ammonium ion have eight valence electrons.
• Ammonia has a trigonal pyramidal molecular geometry. This is because of the lone pair of electrons.
• The Ammonium ion has a tetrahedral molecular geometry since it has four single bonds to the H atoms and no lone pairs.
• Ammonia (NH3) is a single molecule. The Ammonium ion (NH4+) exists bonded to other atoms, like Cl in the compound NH4Cl, or surrounded by water molecules when dissolved in water.
Resources for the Ammonia (NH3):
Lewis Structure: [ Ссылка ]
Molecular Geometry and Bond Angles: [ Ссылка ]
Hybridization: [ Ссылка ]
Formal Charge: [ Ссылка ]
Resources for the Ammonium Ion (NH4+):
Lewis Structure: [ Ссылка ]
Molecular Geometry and Bond Angles: [ Ссылка ]
Hybridization: [ Ссылка ]
Formal Charge: [ Ссылка ]
Other Resources:
Drawing Lewis Structures: [ Ссылка ]
For more practice, see [ Ссылка ]
Finding Valence Electrons: [ Ссылка ]