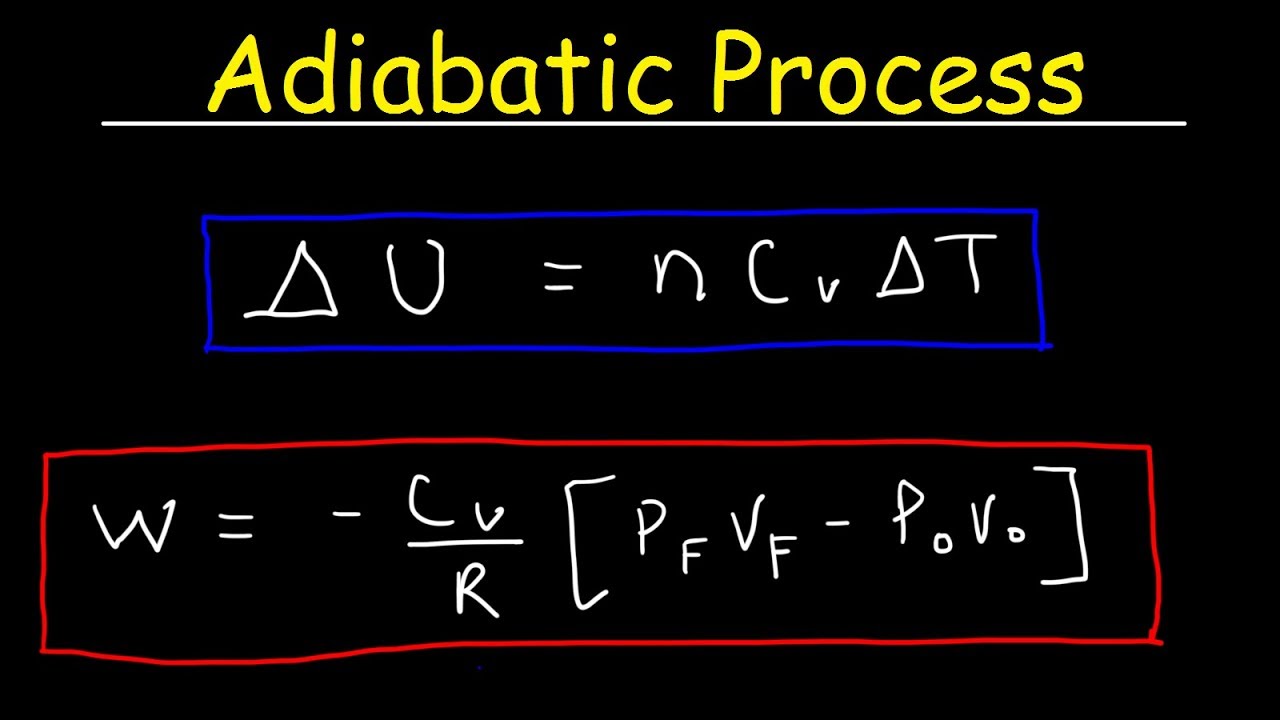
This physics video tutorial provides a basic introduction into adiabatic processes. An adiabatic process occurs when the transfer of heat is equal to zero. Therefore, the work done by a gas under adiabatic expansion is equal to the negative change in the internal energy of the gas. This video explains how to calculate the work done by a gas given the initial and final values of the pressure and volume of the gas. It also explains how to calculate the work done by the gas using the molar heat capacity and the gamma ratio of the gas. This video on thermodynamics contains plenty of examples and practice problems.
Open Vs Closed Vs Isolated System:
[ Ссылка ]
First Law of Thermodynamics:
[ Ссылка ]
Isobaric Process:
[ Ссылка ]
Isochoric Process:
[ Ссылка ]
Isothermal Process:
[ Ссылка ]
Internal Energy of an Ideal Gas:
[ Ссылка ]
_________________________
PV Diagrams:
[ Ссылка ]
Thermodynamics Review:
[ Ссылка ]
2nd Law of Thermodynamics:
[ Ссылка ]
Heat Engines:
[ Ссылка ]
Converting Heat Into Electricity:
[ Ссылка ]
________________________
Carnot Cycle:
[ Ссылка ]
Otto Cycle:
[ Ссылка ]
Refrigerators and Heat Pumps:
[ Ссылка ]
Entropy:
[ Ссылка ]
Heat Engines and Refrigerators Review:
[ Ссылка ]
Physics PDF Worksheets:
[ Ссылка ]









![[REST API на Golang #9] Создание и получение списков](http://i.ytimg.com/vi/KpTZuZE5HS8/mqdefault.jpg)
































































