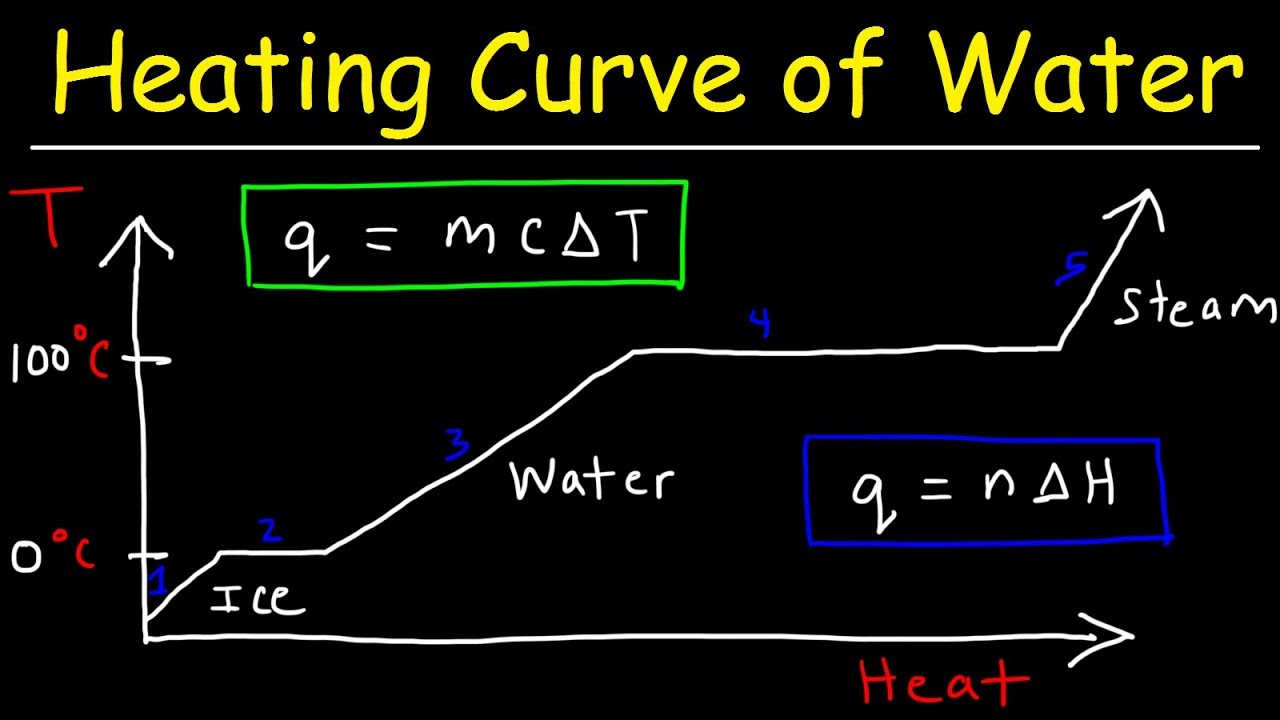
This chemistry video tutorial provides a basic introduction into the heating curve of water and the cooling curve of water. As heat is added to water, the temperature increases which increases the kinetic energy of the molecules. At the freezing point of ice, adding heat will not change the temperature but will increase the potential energy of the molecules as ice melts to liquid water. The enthalpy of fusion is the energy required to melt 1 mole of ice. The enthalpy of vaporization is the energy required to boil 1 mole of water into steam. The slope of the lines is inversely related to the specific heat capacity of the substance. The heating curve represents an endothermic process and the cooling curve is an exothermic process.
Heating Curve of Water: [ Ссылка ]
Heating Curve Chemistry Problems:
[ Ссылка ]
Final Temperature - Ice Water Mixture:
[ Ссылка ]
Molarity, Molality, Density, & Mass %:
[ Ссылка ]
Normality & Equivalent Weight:
[ Ссылка ]
_________________________________
PPM and PPB Concentrations:
[ Ссылка ]
How To Convert PPM to Molarity:
[ Ссылка ]
Enthalpy of Solution & Hydration:
[ Ссылка ]
Solubility Vs Concentration:
[ Ссылка ]
Solubility Curves:
[ Ссылка ]
___________________________________
Henry's Law & Gas Solubility:
[ Ссылка ]
Vapor Pressure & Clausius Equation:
[ Ссылка ]
Raoult's Law - Vapor Pressure:
[ Ссылка ]
Colligative Properties:
[ Ссылка ]
Chemical Kinetics Initial Rate Method:
[ Ссылка ]
_________________________________
Final Exams and Video Playlists:
[ Ссылка ]
Full-Length Videos and Worksheets:
[ Ссылка ]
Chemistry PDF Worksheets:
[ Ссылка ]





























































![📣Дэвид Айк - Ловушка. Глава 1️⃣ из 12. Мы здесь, чтобы вспомнить [Аудиокнига]](https://i.ytimg.com/vi/kn9uX0I2vvE/mqdefault.jpg)












