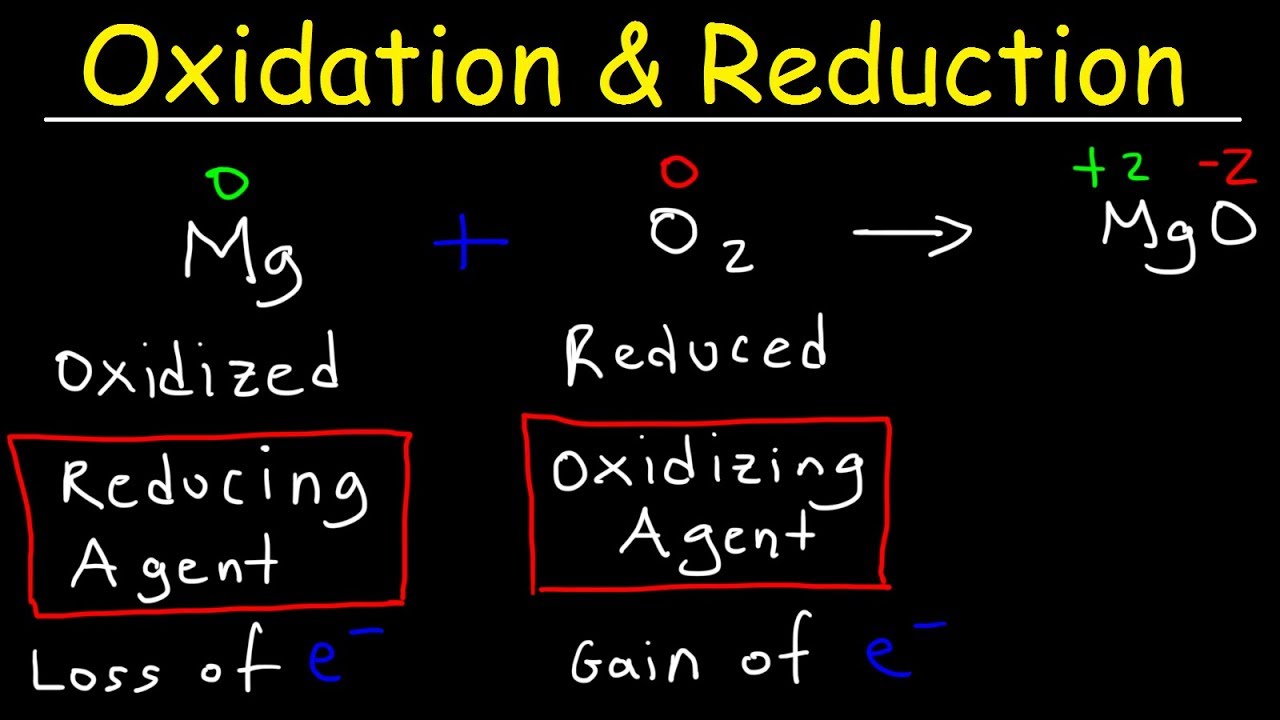
This chemistry video tutorial provides a basic introduction into oxidation reduction reactions also known as redox reactions. This video explains how to identify the substance that is oxidized and reduced. In addition, it explains how to determine which compound or element is the reducing agent and which is the oxidizing agent. Combustion reactions and single replacement reactions are always redox reactions. Double replacement reactions such as precipitation reactions and acid base neutralization reactions are never redox reactions. Combination / Synthesis and Decomposition reactions may or may not be redox reactions.
Full-Length Math & Science Videos:
[ Ссылка ]
___________________________________
Stoichiometry Practice Test:
[ Ссылка ]
Solute, Solvent, & Solution:
[ Ссылка ]
Strong & Weak Electrolytes:
[ Ссылка ]
Molarity Practice Problems:
[ Ссылка ]
Ion Concentration In Solutions:
[ Ссылка ]
Dilution Problems:
[ Ссылка ]
___________________________________
Types of Chemical Reactions:
[ Ссылка ]
Solubility Rules:
[ Ссылка ]
Predicting The Products of Reactions:
[ Ссылка ]
Activity Series of Metals:
[ Ссылка ]
Will This Reaction Occur?
[ Ссылка ]
Predicting Products of SR Reactions:
[ Ссылка ]
___________________________________
Double Replacement Reactions:
[ Ссылка ]
Net Ionic Equations:
[ Ссылка ]
Writing Chemical Equations From Words:
[ Ссылка ]
Solution Stoichiometry:
[ Ссылка ]
Molarity & Dilution Problems:
[ Ссылка ]
Acid Base Neutralization Reactions:
[ Ссылка ]
____________________________________
Acid Base Titration Problems:
[ Ссылка ]
Mixture Problems:
[ Ссылка ]
Calculating Oxidation Numbers:
[ Ссылка ]
Oxidation and Reduction Reactions:
[ Ссылка ]
Balancing Redox Reactions:
[ Ссылка ]
Ideal Gas Law Problems:
[ Ссылка ]
___________________________________
Final Exams and Video Playlists:
[ Ссылка ]